What Did the Inflation Reduction Act (IRA) Change?
And how does it affect value and evidence strategies for biopharma
TL;DR
The IRA is compressing commercial windows for high-spend Medicare drugs, changing how manufacturers approach launch pricing, evidence generation, and portfolio planning.
Impact on biopharma value and evidence strategies:
Greater risk of underpricing at launch
Medicare exposure a bigger factor for pipeline decisions
More reliance on real-world evidence to prove differentiation
Ripple effects likely from competitor negotiations
Small molecules may face more pressure than biologics
Indication sequencing strategies may change in some areas (e.g., oncology)
The Inflation Reduction Act (IRA)
The IRA has introduced one of the biggest shifts in decades for US biopharma.
Before the IRA, price erosion in Medicare was largely driven by market forces: competition, generic entry, and payer negotiations. Manufacturers expected prices to decline over time, but the timing and magnitude varied widely across drugs.
For the first time, the IRA allowed CMS to negotiate Medicare prices directly with manufacturers, resulting in Maximum Fair Prices (MFPs) for selected drugs.
The IRA also introduced other notable changes, including:
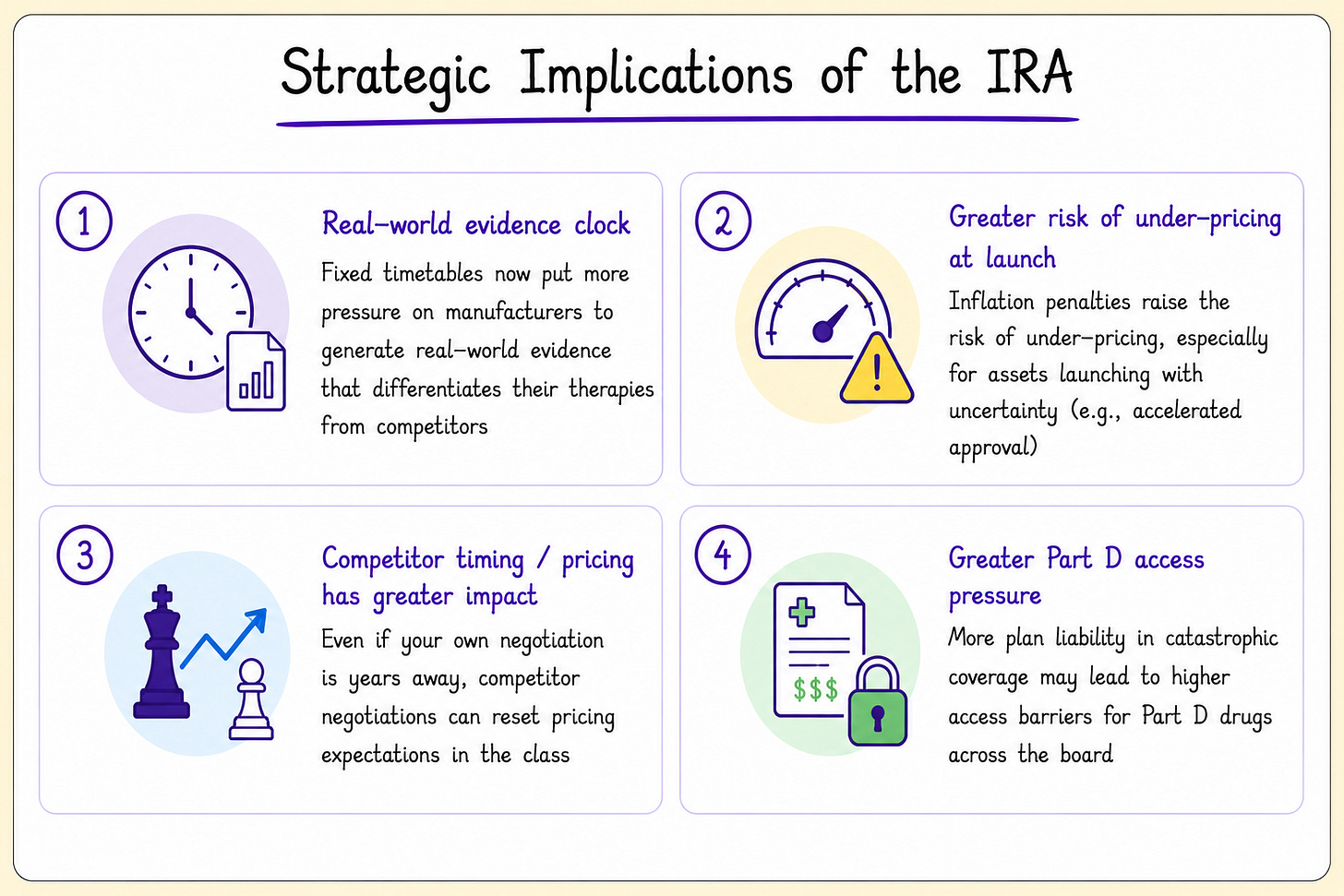
Strategic Implications of the IRA
Early negotiation cycles have shown that CMS is focused on drugs with high Medicare spending and is placing significant weight on a drug’s ability to differentiate itself vs. therapeutic alternatives.
Because head-to-head trials between competitors are often unavailable, manufacturers will increasingly turn to real-world evidence to prove differentiation.
Additionally, manufacturers face new risks if drugs are underpriced at launch, if competitors face earlier IRA negotiations, and if Part D plans increase utilization management and access barriers.
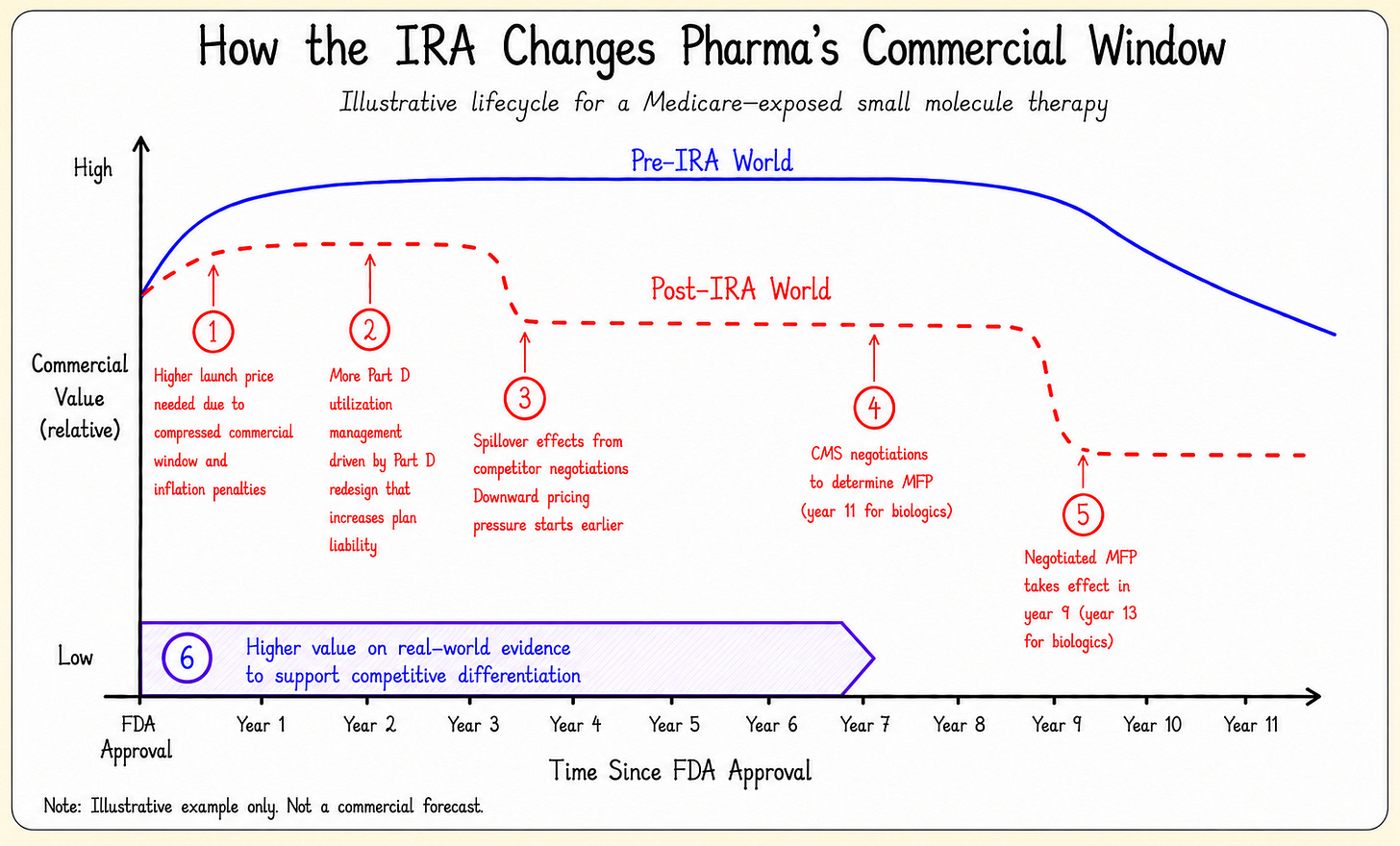
Hypothetical Impact for a Small-Molecule Drug
A hypothetical example is shown below for “Drug X”, a small-molecule drug, to illustrate how these changes might impact commercial value in Medicare.
In early years, more aggressive utilization management by Part D plans may lead to lower sales volumes. By year 3, a competitor’s negotiation could also put downward pricing pressure on Drug X.
By year 7, Drug X might have enough Medicare spend to be selected for negotiation. The negotiated MFP would then be implemented in year 9.
How Does the IRA Change Manufacturer Strategy?
Much discussion of the IRA has focused on CMS’ new ability to negotiate prices. However, this price negotiation is only one piece of the bigger strategic puzzle.
In fact, the IRA impacts many upstream decisions that happen far earlier, such as which assets to prioritize, which competitors to focus on, how to price at launch, what post-launch evidence to generate, and how to sequence indications.
1. Portfolio prioritization and scenario planning
The IRA compresses commercial value for Medicare drugs, and Medicare exposure is now a major factor when forecasting return on investment (ROI). For portfolio planning, this may cause some drugs to be re-prioritized.
Concretely, a promising drug focused on Medicare patients may look less attractive once negotiated prices, competitor timelines, and other factors are modeled. This may be felt more acutely by small molecules given earlier negotiation timelines.
2. Competitor timing and differentiation
The timing of competitor negotiations is also becoming an important factor. If a competitor launches ahead of you, its negotiated MFP may accelerate pricing pressure. Manufacturers that generate evidence to differentiate their therapies vs. competitors may be more sheltered from these spillover effects.
3. Launch pricing
With new inflation penalties, manufacturers are less able to raise post-launch prices. This increases the risk of underpricing, since the effect will be felt for many years. However, overpricing can still lead to payer pushback and more restrictive access.
The challenge is setting a launch price that reflects a drug’s full value while accounting for any residual uncertainty, payer responses, and compressed commercial windows.
4. Evidence generation
CMS negotiations have been focused on how drugs compare vs. alternatives. Since competitor head-to-head trials are often unavailable, manufacturers will rely more on real-world evidence. For drugs likely to be negotiated (or have competitors negotiated), supporting evidence should be planned early to ensure data are generated, analyzed, and published ahead of negotiations.
5. Indication sequencing
The IRA may also change indication sequencing strategies. In treatment areas like oncology, manufacturers often launch first in smaller / later-line populations before expanding to larger / earlier-line populations.
But the clock for CMS negotiations now starts after the first FDA approval. This means that earlier launches in smaller populations may leave less time to capitalize on future expansions in larger populations. These second-order effects may push manufacturers to weigh different market scenarios and rethink indication sequencing strategies.